When the temperature decreases, water converts into solid, and when the temperature increases, water converts into gas. Water is abundantly available in the world and is found in three forms ( solid, liquid, and gas). The density of a substance is usually measured in grams per cubic centimeter. Density of Water Formulaĭensity is calculated by the formula d = M/V, where d is density, M is mass, and V is volume. At normal temperature, water has a density of 0.99823 g/ml. Water does not have any absolute density as the density of water varies with its temperature. Density of distilled water – 0.99823g/mlĭensity of seawater – 1023.6 kg/m 3 at a 25 ☌ temperature, 35 g/kg salinity, and 1 atm pressure.Ībsolute Density is the mass of a substance per unit volume of a material.Density of freshwater – 1g/cm 3 or 1000kg/m 3.Density of pure water – 1g/cm 3 or 1000kg/m 3.Why doesn’t the moon fall into the earth?Īt normal temperature, water’s density is 998.2 kg/m 3. 22° C, the density of water is 997.77 kg/m 3. Typically, in SI units, the density of water at 4☌ is 1000 kg.m - 3. 
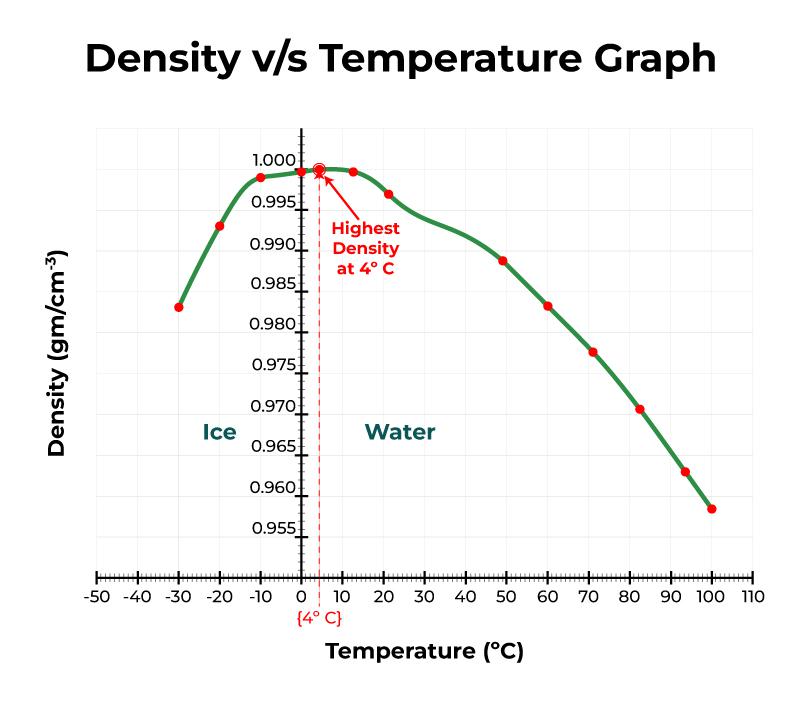
This behavior of water is called “density anomaly”.Above 4 degrees, water’s density decreases as the temperature increases.Unlike normal liquids, the density of water decreases from 4° to 0° Celsius.The density of water is maximum at 4☌ as at this temperature, two opposing effects (thermal expansion and the hydrogen bond formation) are in equilibrium.A molecule of water (H 2O) is made up of two atoms of hydrogen and one atom of oxygen bound by 2 covalent bonds.The densityof a substance can be calculated as its massper unit of volume. Water is an odorless, tasteless, colorless, and transparent chemical substance that is the main constituent of the lakes, oceans, and streams on the planet. The density of water is approximately 1 gram per cubic centimeter (1 gm/cm 3). Density describes how much space an object takes up in relation to its mass.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
